 
They therefore offer a better precision and lower detection limits. Electrodeless discharge lamps are typically much more intense and in some cases, more sensitive than comparable hollow cathode lamps. The energy will vaporize and excite the atoms, inside the bulb, causing them to emit their characteristic spectrum. When power is applied to the RF generator, an RF field is created. The bulb is placed inside a small, self-contained RF generator. A small amount of the metal or salt of the element for which the source is to be used is sealed inside a quartz bulb. 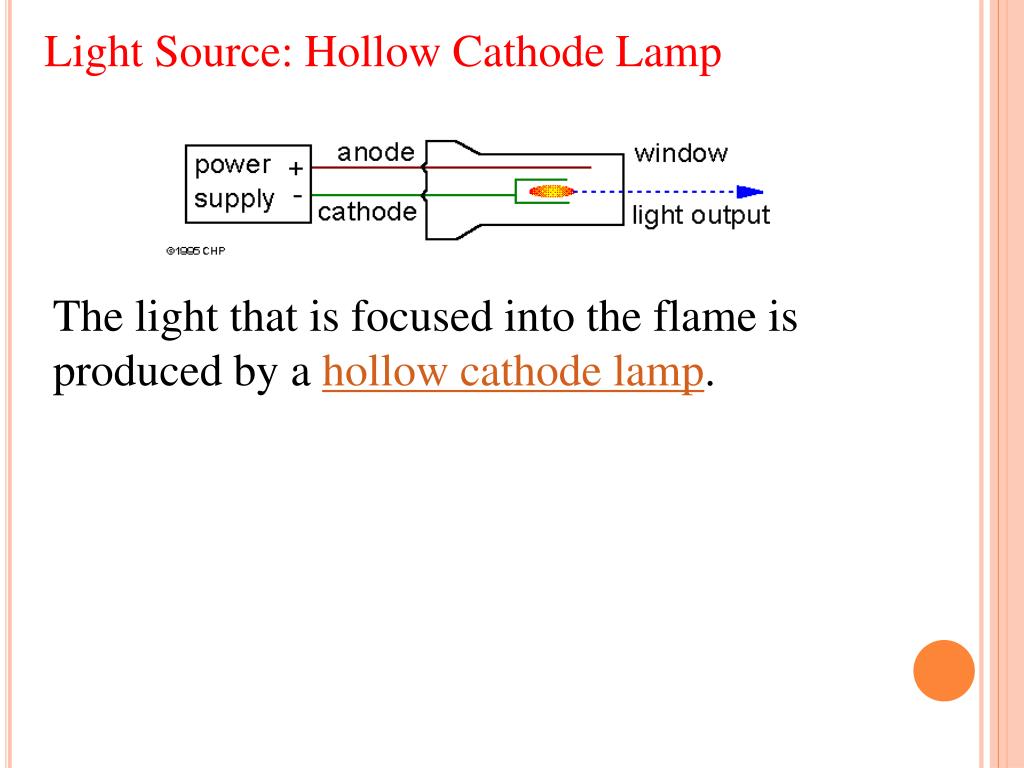
In cases like the above the electrodeless discharge lamp ( EDL) is used. In a few cases, however such as the more volatile elements, the quality of the analysis is impaired by limitations of the hollow cathode lamp. With extended use, the sputtering process removes some of the metal atoms from the cathode and these are deposited elsewhere.įor most elements, the hollow cathode lamp is a completely satisfactory source for atomic absorption. High-intensity hollow cathode lamps (HI-HCLs. HCLs are almost ideal line sources for AAS because of their high stability and narrow linewidth (0.002 nm), but their relatively low intensity is a disadvantage for AFS. Hollow cathode lamps have a finite lifetime. A HCL is composed of a silica envelope that contains 15 Torr of argon or neon and two metal electrodes. The shape of the cathode concentrates the radiation into a beam which passes through a quartz window, and the shape of the lamp is such that most of the sputtered atoms are redeposited on the cathode. Deuterium Lamp to suit a wide range of UV-Vis and AAS Spectrometers, bare D2 lamp and pre-aligned lamp available. Some sputtered atoms are in excited states and emit radiation characteristic of the metal as they fall back to the ground state. Hollow Cathode Lamp 1.5 Inch 2 pin to suit most AAS, 4 pin Agilent Coded lamp, 7 pin Thermo Fisher Coded lamp, 2.0 Inch to suit Perkin Elmer Instruments. These gaseous ions bombard the cathode and eject metal atoms from the cathode in a process called sputtering (Fig. When an electrical potential is applied between the anode and cathode, some of the inert gas atoms are ionized. argon or neon – at a pressure between 1Nm-2 – 5Nm-2. These are sealed in a glass tube filled with an inert gas – e.g. This contains a tungsten anode and a cylindrical hollow cathode made of the element to be determined. The common source of light is a “ hollow cathode lamp”. The two most common line sources used in atomic absorption are the “ hollow cathode lamp, HCL” and the “ electrodeless discharge lamp, EDL”. At the heart of the laser optogalvanic spectroscopy investigations is a Hollow Cathode Lamp (HCL), which is typically composed of an anode, a cathode and a buffer gas (e.g. Narrow line sources not only provide high sensitivity, but also make atomic absorption a very specific analytical technique. In order to measure this narrow light absorption with maximum sensitivity, it is necessary to use a line source, which emits the specific wavelengths which can be absorbed by the atom. It is well known that an atom in its ground state absorbs light at discrete wavelengths (E = h*c/λ). 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
